Factory wholesale Fda Approved Mycotoxin Testing - Elisa Test Kit of AMOZ – kwinbon
Factory wholesale Fda Approved Mycotoxin Testing - Elisa Test Kit of AMOZ – kwinbon Detail:
2.The nitrofuran drugs furaltadone, nitrofurantoin and nitrofurazone were banned from use in food animal production in the EU in 1993, and the use of furazolidone was prohibited in 1995. The analysis of nitrofuran drugs residue needs to be based on the detection of the tissue bound metabolites of the nitrofuran parent drugs, since the parent drugs are very rapidly metabolized, and the tissue bound nitrofuran metabolites will retain for a long time, therefore the metabolites are used as the target in the detection of the abuse of nitrofurans. Furazolidone metabolite (AMOZ), Furaltadone metabolite (AMOZ), Nitrofurantoin metabolite (AHD) and Nitrofurazone metabolite (SEM).
Details
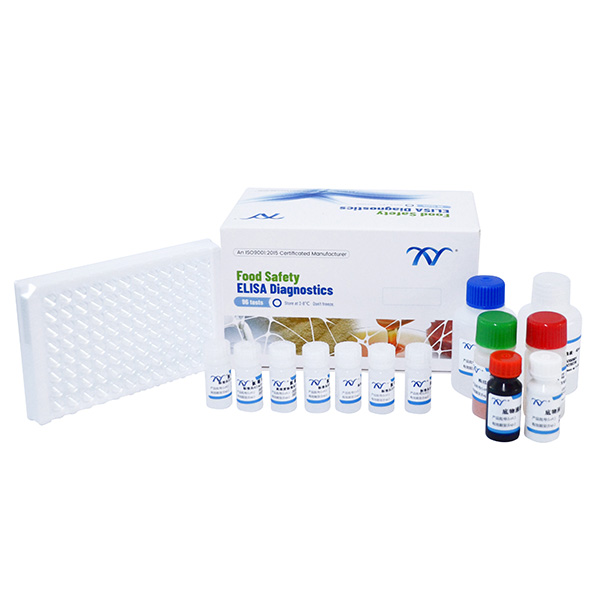
1.Elisa Test Kit of AMOZ
2.Cat. KA00205H-96 Wells
3.Kit Components
● Microtiter plate with 96 wells coated with antigen
● Standard solutions(6 bottles)
0ppb, 0.05ppb,0.15ppb,0.45ppb,1.35ppb,4.05ppb
● Spiking standard solution: (1ml/bottle) ……………………………………………...100ppb
● Enzyme conjugate 1ml…………………………………………………..…………….red cap
● Antibody solution 7ml ………………………………………………..………….….green cap
● solution A 7ml…………………………………………………….…………………white cap
● solution B 7ml………………………………………………………….………..…… red cap
● stop solution 7ml ……………………………………………………….…………yellow cap
● 20×concentrated wash solution 40ml…………………………………….……transparent cap
● 2×concentrated extraction solution 50ml……………………………………….…….blue cap
● 2-Nitrobenzaldehyde 15.1mg………………………………………………….……white cap
4.Sensitivity, accuracy and precision
Sensitivity: 0.05ppb
Detection limit
Aquatic products(fish and shrimp)………….……… 0.1ppb
Accuracy
Aquatic products(fish and shrimp)….…...………… 95±25%
Precision: CV of the ELISA kit is less than 10%.
5.Cross Rate
Furaltadone metabolite(AMOZ)………………………………….…………100%
Furazolidone metabolite(AMOZ)…………………………..………………..<0.1%
Nitrofurantoin metabolite(AHD)……………………………….……………<0.1%
Nitrofurazone metabolite(SEM)…………………………………………..…<0.1%
Furaltadone…………………………………………………………….…….11.1%
Furazolidone……………………………………………………….….…..…<0.1%
Nitrofurantoin……………………………………………………….…….…<1%
Nitrofurazone…………………………………………….………………..…<1%
Product detail pictures:

Related Product Guide:
With sophisticated technologies and facilities, strict top quality handle, reasonable value, exceptional support and close co-operation with clients, we are devoted to furnishing the ideal worth for our clients for Factory wholesale Fda Approved Mycotoxin Testing - Elisa Test Kit of AMOZ – kwinbon , The product will supply to all over the world, such as: Johor, Netherlands, Mauritius, In order to make more people know our products and to enlarge our market, we have devoted a lot of attention to technical innovations and improvement, as well as replacement of equipment. Last but not the least, we also pay more attention to training our managerial personnel, technicians and workers in planned way.
The company keeps to the operation concept "scientific management, high quality and efficiency primacy, customer supreme", we have always maintained business cooperation. Work with you,we feel easy!